The overall energy of this nucleus would be reduced if a proton could somehow transform itself into a neutron. Its filled energy levels would look like the well on the left. Well, nature allows this transformation and we call it - decay!Īs another example, consider 18F, which consists of 9 neutrons and 9 protons. The overall energy of the nucleus would be reduced (and its stability increased) if the “stray” neutron at the top of the neutron well could somehow transform itself into a proton and jump down to the lower energy state in the proton well. For example, silver-106 undergoes electron capture to become palladium-106. The different types of decay are alpha, beta, positron emission, electron capture, gamma emission, and spontaneous fission. 
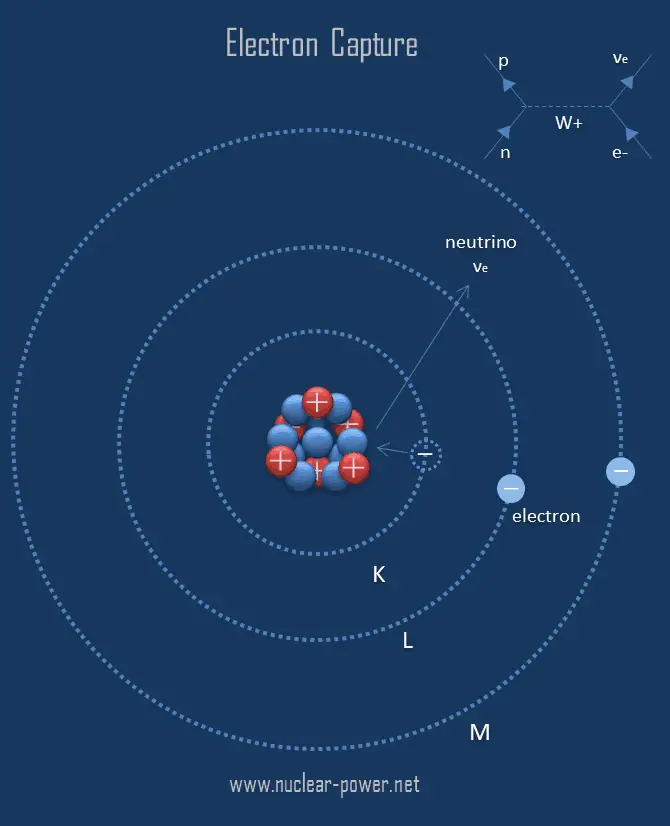
Figure 20.3.1: Common Modes of Nuclear Decay. In electron capture, an electron from an inner orbital is captured by the nucleus of the atom and combined with a proton to form a neutron. The most common are alpha and beta decay and gamma emission, but the others are essential to an understanding of nuclear decay reactions. The filled energy levels would look like the well on the left. An alternate way for a nuclide to increase its neutron to proton ratio is by a phenomenon called electron capture, sympolized E.C. Both beta-plus and beta-minus, if allowed, always dominate electron capture since electron capture involves the relatively rare occurrence of a sizable overlap between electron and proton wavefunctions.īeta decay can be understood conceptually by looking carefully at the differences in the potential wells for protons and neutrons, and the order in which the available energy levels are filled.įor example, consider 24Na, which consists of 13 neutrons and 11 protons. The exception to this rule involves electron capture. If more than one decay involves a positive Q, the one that releases the most energy will typically dominate. Therefore, 81Kr will decay via electron capture, and release 0.281 MeV of energy per decay. (a) What other particle or particles are emitted in the decay (b) Assume that the electron is captured from the K shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
